Home » Keratoconus
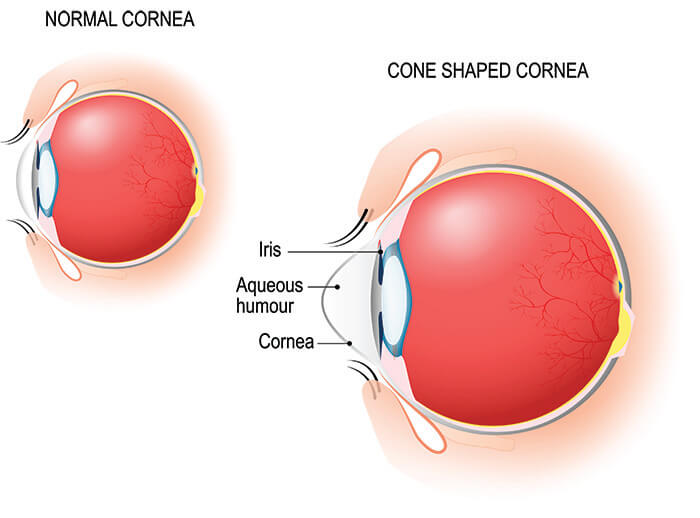
Some patients with very high levels of astigmatism and nearsightedness may have an eye disease called keratoconus. Keratoconus affects the cornea – the clear lens in front of your iris. Keratoconus is caused by abnormal collagen within the cornea and results in an irregular or warped curvature of the cornea. This in turn degrades the focus of the eye. In the past keratoconus could be very challenging to treat. Fortunately the development of new procedures to treat keratoconus has improved the prospect for maintaining vision or even making it better.
How Do I Know I Have Keratoconus?
Keratoconus is a relatively uncommon condition affecting less than 1% of people. Most patients with astigmatism or nearsightedness do not have keratoconus. However if you have high or increasing levels of astigmatism and very frequent prescription changes or if you have a family history of keratoconus or poor vision with glasses or contacts you should have an eye exam.
Patients with keratoconus often seek out LASIK surgeons in the hope that laser eye surgery will correct their vision problem. Some patients with keratoconus are unaware that they have the condition. However patients with keratoconus should generally not have LASIK because LASIK can actually make the condition worse. Because we encounter on a regular basis patients with keratoconus, Sun Laser Vision Center has made it a priority to specialize in helping patients with the condition.
Certain risk factors can increase the potential for developing keratoconus. The most important of these is a family history of keratoconus. Patients who frequently rub their eyes or who have certain types of allergic conditions such as hay fever or eczema may be at greater risk of developing keratoconus. People with Down’s syndrome also have higher rates of keratoconus.
Keratoconus is a progressive disease. The earlier it is diagnosed and treated the better the possibility of having lifelong good functional vision. If you think you have keratoconus or if you have a family member with keratoconus don’t wait. We recommend that you get evaluated today. Please contact Sun Laser Vision Center to schedule your exam.
What Are the Symptoms of Keratoconus?
Symptoms Linked to Keratoconus Include:
- Poor focus even with glasses or contacts
- Distorted vision
- Blurry vision
- Light and glare sensitivity
- Difficulty with night driving
- Frequent lens prescription changes
- Cloudy vision
- Sudden worsening of vision
How Does My Doctor Diagnose Keratoconus?
To find out if you have keratoconus you will need to have a full dilated eye exam with measurements of your vision, your glasses prescription and imaging tests of your cornea. Your ophthalmologist will complete the eye exam and review your medical history and results of your tests. Patients with keratoconus often have thin corneas with a high or irregular curvature of the cornea. Some patients may also have characteristic scars within the cornea.
How is Keratoconus Treated?
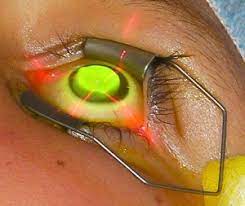
Corneal Cross Linking (CXL)
Corneal cross-linking is a procedure that has been newly approved by the FDA. Initially conceptualized in the late 1990s, corneal cross-linking has become the standard of care to treat patients with keratoconus in its early stages. The best way to preserve vision in patients with keratoconus is to ensure that the condition never becomes severe in the first place.
Corneal cross-linking is intended to stabilize a cornea with keratoconus. It does not necessarily improve vision. Therefore early detection and diagnosis are critical. Corneal cross-linking has proven to be highly effective with a success rate of 90% to 95%.
Corneal cross-linking is performed by gently removing the protective barrier cells on the surface of the cornea and then placing drops of a riboflavin solution onto the cornea. Riboflavin is an essential vitamin in our diets. Riboflavin has the unique characteristic that when dissolved in water it can absorb light energy in the UV spectrum. After placing riboflavin drops for 30 minutes on the cornea a UV light is turned on and the riboflavin will absorb the UV energy and in turn strengthen the molecular bonds of collagen in the cornea. This can increase the strength of the cornea by up to 300%. This particular technique is what is known as “epi-off” (epithelium removed). This is the technique that we use at Sun Laser Vision Center.
There are modifications to the corneal cross-linking technique which we use at Sun Laser Vision Center. We have adopted these modifications because based on scientific literature and data these appear to be more beneficial to patients. Not all of these techniques and not all of the instruments used in cross-linking are approved by the FDA. When techniques, instruments or devices that are outside of the parameters of the original FDA study are used, we call this off-label or non-label.
Dr. Foote was the first ophthalmologist to perform corneal cross-linking. We began offering the procedure in 2014. Contact Sun Laser Vision Center today to request an appointment for an exam and determine if cross-linking may help you.
Intacs
Intacs are semicircular rings of Plexiglas which can be placed within a cornea. Originally designed as an alternative to LASIK, Intacs were approved by the FDA through what is called a compassionate device exemption to help treat patients with keratoconus.
Intacs increase the thickness of the cornea and can change the curvature the cornea reducing or regularizing astigmatism within the cornea. They have proven to be both safe and effective in helping to improve vision in patients with keratoconus.
Intacs are placed within the cornea using the same femtosecond laser that is used in all laser LASIK. The laser will create a channel in which the intac segment is placed. An operating microscope is used to help assist with placing the intac. It is placed outside of the line of sight and does not directly focus light itself. Instead the intac will change the curvature of the cornea to improve the vision.
Intacs are placed either as a single semi-circle or as paired semi-circles. The procedure is generally comfortable for most patients to go through and has a fairly quick recovery. Usually, within 1 to 3 days patients can return to most of their regular activities. Patients will use prescription eye drops during the recovery period.
Intacs and cross-linking can be combined in patients with keratoconus. It is not advisable to perform both procedures at the same time, however. Usually, a waiting period of 3 to 6 months is recommended to ensure that the cornea fully heals from one procedure before performing the second.
The main purpose of placing Intacs is to improve the curvature and improve the quality of vision for patients with keratoconus. Intacs are generally considered when patients have mild to moderate keratoconus and there is reasonable potential for improving vision. Keep in mind that in patients with very advanced keratoconus Intacs may not be recommended if the amount of correction is too small to make an effective difference for patients.
Sun Laser Vision Center has been performing the placement of Intacs since 2008.
EVO ICL
The EVO ICL is an implantable device that can be placed inside the eye behind the Iris. The ICL is a corrective lens made of collagen and synthetic polymer. The ICL can correct very high degrees of nearsightedness and moderate degrees of astigmatism. Patients with moderate keratoconus who have relatively regular astigmatism may be candidates for placement of ICL.
The ICL may be able to achieve improved vision for patients with very high degrees of nearsightedness over what can be obtained with glasses. For patients with keratoconus who are not tolerant of contact lens wear, ICL may be a significant benefit in improving their vision.
Please refer to the section on vision correction surgery on this website for additional information about ICL and for links to Staar Surgical, the manufacturer of the EVO ICL. Please note that EVO ICL is FDA approved for the treatment of myopia with or without astigmatism. Studies evaluating the use of ICL in patients with keratoconus have not been submitted to the FDA. Use of ICL in patients with keratoconus is considered off-label.
Dr. Foote was the first surgeon to perform ICL surgery in patients with keratoconus in the El Paso region. At Sun Laser Vision Center we have extensive experience in placing the ICL.
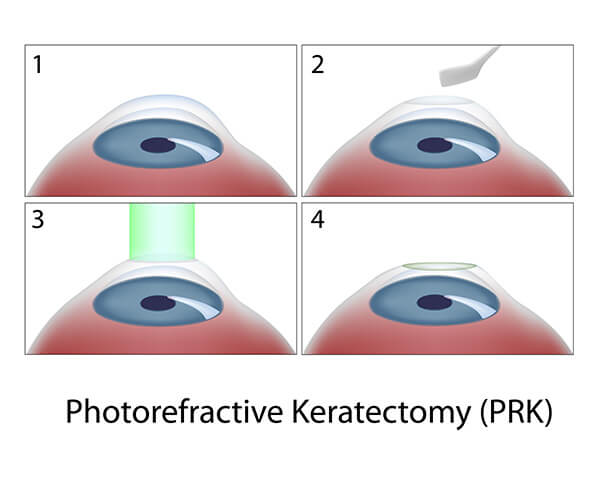
PRK
Patients with keratoconus who have low degrees of nearsightedness and or astigmatism can in certain circumstances be candidates for PRK combined with cross-linking or for patients who had previously undergone cross-linking. PRK as a treatment for keratoconus can sometimes improve the best potential vision.
Use of PRK in patients with keratoconus can be an effective treatment for improving vision however there is some underlying risk that it could worsen keratoconus as well. Generally this risk is mitigated by performing simultaneous or sequential corneal cross-linking. Please be aware that PRK is not FDA approved for the treatment of keratoconus, but has been fairly extensively studied outside of the FDA process and reported in the medical literature to be effective in select patients with mild degrees of keratoconus.
Corneal transplant
Partial or complete transplantation of the cornea may be necessary in patients who have very advanced degrees of keratoconus which cannot be corrected with contact lenses or by any of the surgical options listed above. It may also be necessary in patients who have extensive scarring as a result of keratoconus and have non-correctable vision.
Corneal transplantation as a technique has been performed for over 40 years. It may be the only viable treatment option for patients with advanced keratoconus. The risk benefits and alternatives to corneal transplantation will be extensively discussed with those patients who have advanced keratoconus.
Click the button below to request an appointment today.
LEARN MORENonsurgical options
Contact lenses are often the best option for focusing eyesight in patients with keratoconus. As opposed to spectacles, contact lenses effectively create a new curvature of the cornea when they are placed on the surface. For that reason contacts provide a better focus for patients with keratoconus who have abnormal curvature of the cornea.
Depending on the severity of keratoconus the type of contact lens we recommend may change. For example patients with very mild keratoconus may be able to use regular soft contact lenses or soft contact lenses with astigmatism correction. Patients with moderate degrees of keratoconus may require a hard contact lens or hybrid lens which is hard in the center and has a soft skirt. Patients with more advanced keratoconus or very irregular curvatures or irregular astigmatism may require a larger contact lens called a scleral lens. Scleral contact lenses vault over the cornea and actually rest just outside of the cornea on the white part of the eye called the sclera.